Fuel
Formula One cars use gasoline as a fuel for their engines. For most of the teams, these fuels are supplied for free by a technical partner that is usually a worldwide manufacturer. Fuel (in this case gasoline) is the liquid that is mixed with air into the cylinder to generate a combustion, and therefore a powerful expansion in the cylinder. The better the fuel can be distributed in the combustion chamber and the more explosive particles there are per cc (cm³) in the fuel, the more power you will be able to produce with a constant amount of fuel, or the higher the fuel economy will be.
It would be easy if there were no restrictions. The International Sports Car Federation (FIA) restrictions include the octane count, oxygen and nitrogen content and density. Gasoline cannot contain compounds that are not found in commercial gasoline. These requirements are based on European Parliament decisions for commercial gasoline produced since 2000. Thanks to mathematical models for predicting the power and end characteristic of the gasoline, a fuel can be prepared to increase the power in cars within the restrictions set by the FIA.
Specifications
Back in the founding years of Formula 1, the mixture was left up to the chemists. They brewed aggressive mixtures from substances such as benzene, methanol, acetone and nitrobenzene, some of which had to be drained from the engine immediately after practice and races. Without this precaution, the engine would not have survived the night. Later on, kerosene was used until the late 1960s, before the list of admissible additives was increasingly narrowed by the sport's governing body, the FIA, for the protection of drivers and mechanics.
By the end of the 1970s, regulations provided for the use of high-octane gasoline, on sale in France, Italy, Germany and England. Gasoline with RON 101 available at European gas stations was used, with tolerance of up to RON 102. When this kind of gasoline was no longer for sale, the team suppliers were allowed to prepare special fuels for the category. They, therefore, became more elaborate and differed widely from commercial gasoline.
At the end of the 1980s the RON 102 restraint continued, and restrictions were now applied to oxygen and nitrogen content (2% in both cases). As time went by, new restraints were established and others, such as steam pressure, density, benzene and lead content, were added. The petroleum companies experimented with more than 300 mixtures per season up until the early 1990s.
In 1992, the FIA decided it would be illegal to use any substance in fuel that was not found in commercial gasoline. Today and since 1993, only unleaded 'super' petrol compliant with EU standards is permitted, the same petrol available at the service station around the corner.
Production
 Production begins for the purpose of obtaining as much power as possible from the engine without jeopardizing durability and with focus on fuel consumption. The first stage covers the use of a computer forecast model, in which a database is fed with more than a hundred raw materials to obtain experimental gasoline. In their search for the best mixture and the extra shot of power for those crucial split seconds, the engineers can mix and burn petrol virtually, without actually having to produce the fuel. Some of the teams have developed their computer technology so that the virtual petrol can even be tested in a virtual engine. This mathematical model was designed using the Formula 1 engine response to different kinds of fuel and all physicochemical properties of the raw materials.
Production begins for the purpose of obtaining as much power as possible from the engine without jeopardizing durability and with focus on fuel consumption. The first stage covers the use of a computer forecast model, in which a database is fed with more than a hundred raw materials to obtain experimental gasoline. In their search for the best mixture and the extra shot of power for those crucial split seconds, the engineers can mix and burn petrol virtually, without actually having to produce the fuel. Some of the teams have developed their computer technology so that the virtual petrol can even be tested in a virtual engine. This mathematical model was designed using the Formula 1 engine response to different kinds of fuel and all physicochemical properties of the raw materials.
The second stage involves preparing small batches of different kinds of experimental gasoline and testing them in a laboratory to confirm that they meet the specification restrictions, and then are tested in an engine where power and consumption levels are checked. After selecting some fuels that are likely to be successful in the team's engines, durability tests are performed on a dynamometer and on the track. Once the gasoline composition has been decided, a sample is sent to the FIA laboratory in England for approval.
Oil companies produce the lubricants at fore set quantities, with Elf producing batches in quantities of 40m³ (40,000 litres) at Elf’s Solaize research centre and are transported in 50 litre barrels.
A top team with a works supplier takes around 3600 litres of fuel, 200 litres of engine oil, 180 litres of gearbox oil, 80 litres of hydraulic fluid, 20 litres of coolant and several kilos of grease to a race. There is always an engineer present at the race location 5 days in advance to check if all fuels are at location and to control the chromatography and spectrometry machines that allow to conduct engine diagnostics. At the end of a race weekend, all unused products are systematically returned to the manufacturer.
Composition
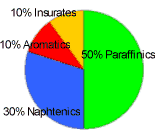
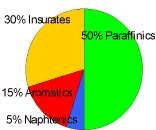
As mentioned before, race fuels in F1 must have the same components as consumer fuels. It is the proportion of the different components that make all the difference. Because of that limitation, the available hydrocarbons can be categorized in two main categories:
- Saturated: In this case, molecules contain enough hydrogen atoms to saturate the carbon atoms.
- Paraffin (straight chains with simple links): High molecular weight alkane hydrocarbons with the general formula CnH2n+2, where n is between 22 and 27. The name is derived from Latin para+affinis meaning here "lacking affinity".
It is mostly found as a white, odorless, tasteless, waxy solid, with a melting point between 47°C and 65°C. It is insoluble in water, but soluble in benzene. Paraffin is unaffected by most common chemical reagents but oxidizes readily. - Naphthenic (closed chains with simple links): These have excellent low temperature properties, mainly used in lubricants.
- Paraffin (straight chains with simple links): High molecular weight alkane hydrocarbons with the general formula CnH2n+2, where n is between 22 and 27. The name is derived from Latin para+affinis meaning here "lacking affinity".
- Non-saturated: In this case, the molecules do not contain enough hydrogen atoms to saturate the carbon atoms.
- Aromatics (closed chain, with double and simple alternating links, that is benzene nucleus): These are so called because of their perfumed smell. The main aromatics are benzene, toluene and the xylenes.
- Diolefins or Dienes (straight chains with two double links).
- Acetylene (straight chains with triple link): Acetylenes are part of the alkynes
| Normal fuel | F1 race fuel |
 |
 |
Fuel tank
 F1 cars use deformable fuel tanks made from puncture-proof Kevlar. This dramatically reduces the risk of any fire during an accident. The tank reshapes itself in the area where any pressure is being put. All the fuel lines are auto cut-off lines. This is incase there is any breakage. There is also a extinguisher placed in the sensitive zone of the car which will automatically turn on in the case of a crash.
F1 cars use deformable fuel tanks made from puncture-proof Kevlar. This dramatically reduces the risk of any fire during an accident. The tank reshapes itself in the area where any pressure is being put. All the fuel lines are auto cut-off lines. This is incase there is any breakage. There is also a extinguisher placed in the sensitive zone of the car which will automatically turn on in the case of a crash.
The size of a fuel tank is also know to be rather important towards chassis design. The size of the tank must be considered against fuel consumption, expected average stint length, aerodynamics, etc. Since the tank is located under and behind the driver's seat, it largely determines the space in between the driver and the engine. The further the sidepods and the driver can be moved back, the cleaner the air will be onto the sidepods. It is therefore clear that for aerodynamic purposes, a tank should be made as small as possible. The same statement also clarifies the importance of fuel economy besides the shorter pitstops.
In current Formula One racecars, fuel consumption can reach 180-200 litres in a 300 km race distance. Drivers can slightly control this number on their steering wheel and can drive relatively economical when performance is not a high priority, for example behind a safety car.
Refuelling
Since the year 1994, refuelling during pit stops is back in Formula One, at that time brought back to increase the excitement for spectators. At some teams there are always three people available to refuel the car. The first one is the 'hoseman' who actually pushes the hose into the car and pulls it out when there is enough fuel into the car. When that moment arrives can be read from a display on the refuelling hose. A second crew member holds the hose right after the hoseman to help him pull the hose away quickly when the refuelling has completed. A third sometimes protects the hot exhausts from the refuelling people, just to take care of safety.
 |
 |
Because a car can be refuelled at a rate of 11 litres per second, many were sceptic in 1994. During the years however, refuelling has become accepted again in F1 and is an integral part of a good pit stop. Accidents have happened during all these years, but to take care of safety as much as possible, the FIA orders a French firm to build these installations to their wishes. Each one of them costs around 30000 EUR. Whenever there is a problem, the fuel line gets cut off by the machinery to limit the danger of fire or an explosion.
Since however performance is all that matters, teams now cover the installations with isolation foil to prevent the fuel to heat up. The lower its temperature, the denser it is, so the more power the engine gets when a constant volume of fuel is injected in each cylinder. Each teams cools its fuel therefore to around 10°C.
